 According to him, they are indestructible and indivisible.Dalton's atomic theory states that all substances consist of building blocks known as atoms.Dalton’s Atomic Theory was proposed by John Dalton in the year 1808.Atoms can be annihilated and changed over into energy.Atoms can consolidate in any proportion and not simply entire numbers. 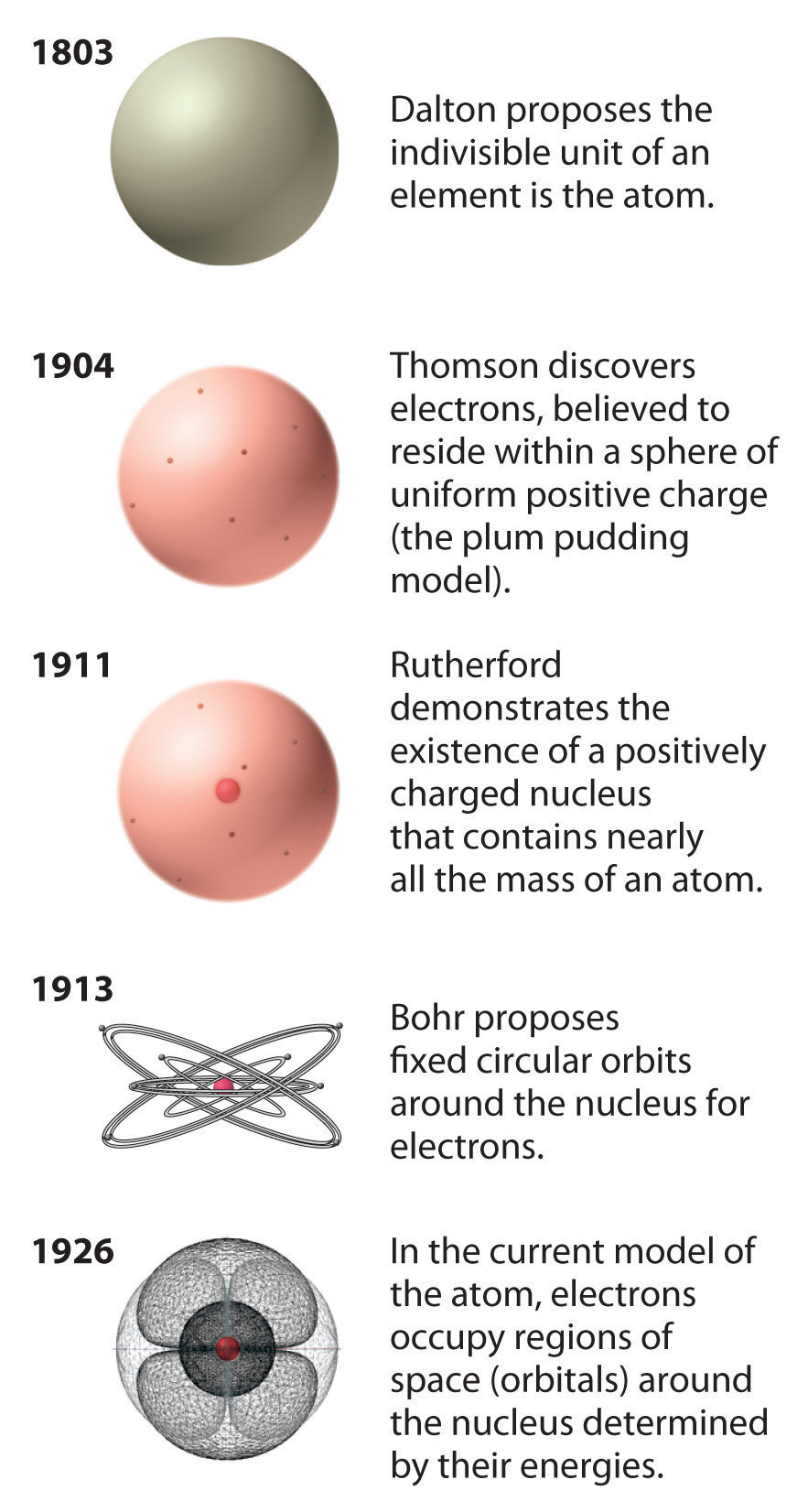
Atoms of various components have similar properties such particles are isobars.Atoms of a similar component have various substances such particles are isotopes.Atoms are dissoluble in protons, neutrons, and electrons.Henceforth, Dalton's Atomic Model, in its most flawless structure, is presently just thought to be substantial for chemical reactions. The Atomic hypothesis was altered with the commitment of numerous get-togethers, subsequently, the a dvanced nuclear hypothesis repudiates Dalton's atomic hypothesis. The theory forms a base to differentiate between elements and compounds.ĭalton’s Atomic Theory vs Modern Atomic Theory.The law of conservation of mass, the law of multiple proportions, and the law of constant proportions are not violated by Dalton’s atomic theory.The following are the merits of Dalton’s Atomic Theory The theory does not account for allotropesĭalton's atomic theory cannot explain the variations in characteristics between diamond and graphite, both of which are composed entirely of carbon. For example, sugar/sucrose (C 11H 22O 11). To form compounds, elements need not combine in simple, whole-number ratiosĬertain complex organic compounds do not have simple ratios of component atoms. However, various isotopes of the same element have different atomic masses. It does not account for isotopesĪccording to Dalton's atomic theory, all atoms of an element have the same mass and density. However, it is possible for two distinct elements to have the same mass number. It does not account for isobarsĪccording to this theory, the masses of atoms from various elements must be different. However, the discovery of subatomic particles (such as protons, electrons, and neutrons) contradicted this theory. The following are the Drawbacks of Dalton’s Atomic Theory It does not account for subatomic particlesĭalton's atomic theory held that atoms were indivisible. Postulate 5: Atoms can be reorganized, united, or isolated in compound responses. 
Postulate 4: Particles of various components can join with one another in fixed entire number proportions to frame compounds.Besides, iotas can't be partitioned into more modest particles. Postulate 3: Atoms cannot be made nor annihilated.Nonetheless, particles of various components display various properties and differ in mass and size. Postulate 2: All iotas of a particular component are indistinguishable in mass, size, and different properties.Postulate 1: All matter is comprised of little, indissoluble particles called atoms.The following are the postulates of Dalton’s atomic theory 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
